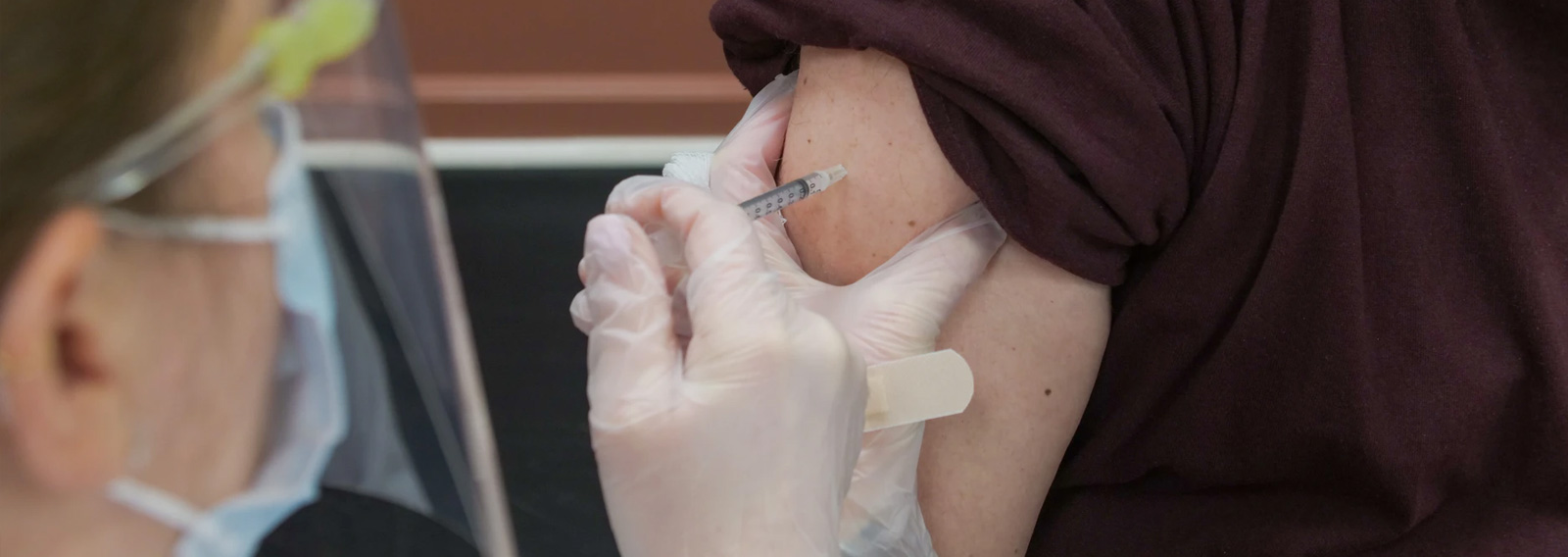
Senator Matthew Canavan has said Australia should halt the rollout of the AstraZeneca vaccine after widespread safety concerns across Europe.
Posting to Facebook and Twitter on Tuesday, Senator Canavan said the nation should not blindly rush ahead with the vaccine when there is no imminent threat of the virus.
“We should pause the rollout of the AstraZeneca vaccine because almost every European country now has concerns over its safety,” he said.
Adding, “There is no imminent threat of coronavirus here so why would we blindly rush on when others are concerned?”
We should pause the rollout of the AstraZeneca vaccine because almost every European country now has concerns over its safety. There is no imminent threat of coronavirus here so why would we blindly rush on when others are concerned?
— Matthew Canavan (@mattjcan) March 15, 2021
The Senator also tweeted a list of countries that have suspended the use of the AstraZeneca vaccine as a precaution following reports that people who received it went on to form blood clots.
These countries include:
- Denmark
- Norway
- Ireland
- Estonia
- The Netherlands
- Thailand
- Bulgaria
- Iceland
- Lithuania
- Luxembourg
- Latvia
- Germany
- France
- Italy
- Spain
- Cyprus
- Portugal
- Slovenia
Despite the concerns overseas, the Australian health advisors and the government remain committed to the rollout of the AstraZeneca vaccine.
According to ABC News, Treasurer Josh Frydenberg said on Tuesday morning there was no evidence yet to confirm a link between the vaccine and the clotting.
“The European equivalent of the TGA [Australia’s Therapeutic Goods Administration] as well as the World Health Organization have said the AstraZeneca vaccine is effective, and there’s no evidence of causation between the vaccine itself and the blood clots,” he told Radio National.
Senator Canavan went on to link a release from the European Medicines Agency, which said a safety committee will review the available information before holding a meeting on Thursday, March 18, “to conclude on the information gathered and any further actions that may need to be taken.”
Senator Canavan added, “There will be further information from authorities on Thursday this week. What is the problem of pausing the rollout for a few days while we wait for this information?”